Determination of Fe, Ni, Cu, Zn, Cd and Pb in seawater by isotope dilution automatic solid-phase extraction—ICP-MS
-
Abstract: A thorough understanding of the biogeochemical cycling of trace metals in the ocean is crucial because of the important role these elements play in regulating metabolism in marine biotas and thus, the climate. However, a precise and accurate analysis of trace metals in seawater is difficult because they are present at extremely low concentrations in a high salt matrix. In this study, we report an analytical method for the preconcentration and separation of six trace metals, Fe, Ni, Cu, Zn, Cd and Pb, in seawater using a seaFAST automatic solid-phase extraction device, analysis by a triple quadrupole collision/reaction technique with inductively coupled plasma mass spectrometry (ICP-MS), and quantification by the isotope dilution technique. A small volume (10 mL) of seawater sample was mixed with a multi-element isotope spike and subjected to seaFAST procedures. The preconcentrate solution was then analyzed using the optimized collision/reaction cell mode of ICP-MS, with NH3 gas for Fe and Cd with a flow rate of 0.22 mL/min and He for Ni, Cu, Zn and Pb with a flow rate of 4.0 mL/min. The procedure blanks were 130 pmol/L, 3.0 pmol/L, 6.8 pmol/L, 37 pmol/L, 0.29 pmol/L and 0.42 pmol/L, for Fe, Ni, Cu, Zn, Cd and Pb, respectively. The method was validated using five reference materials (SLRs-6, SLEW-3, CASS-6, NASS-7 and GEOTRACE-GSC), and our results were consistent with the consensus values. The method was further validated by measuring full-water-column seawater samples from the subtropical Northwest Pacific Ocean, and our results demonstrated good oceanic consistency.
-
Key words:
- seaFAST /
- trace metals /
- isotope dilution /
- seawater /
- ICP-MS
-
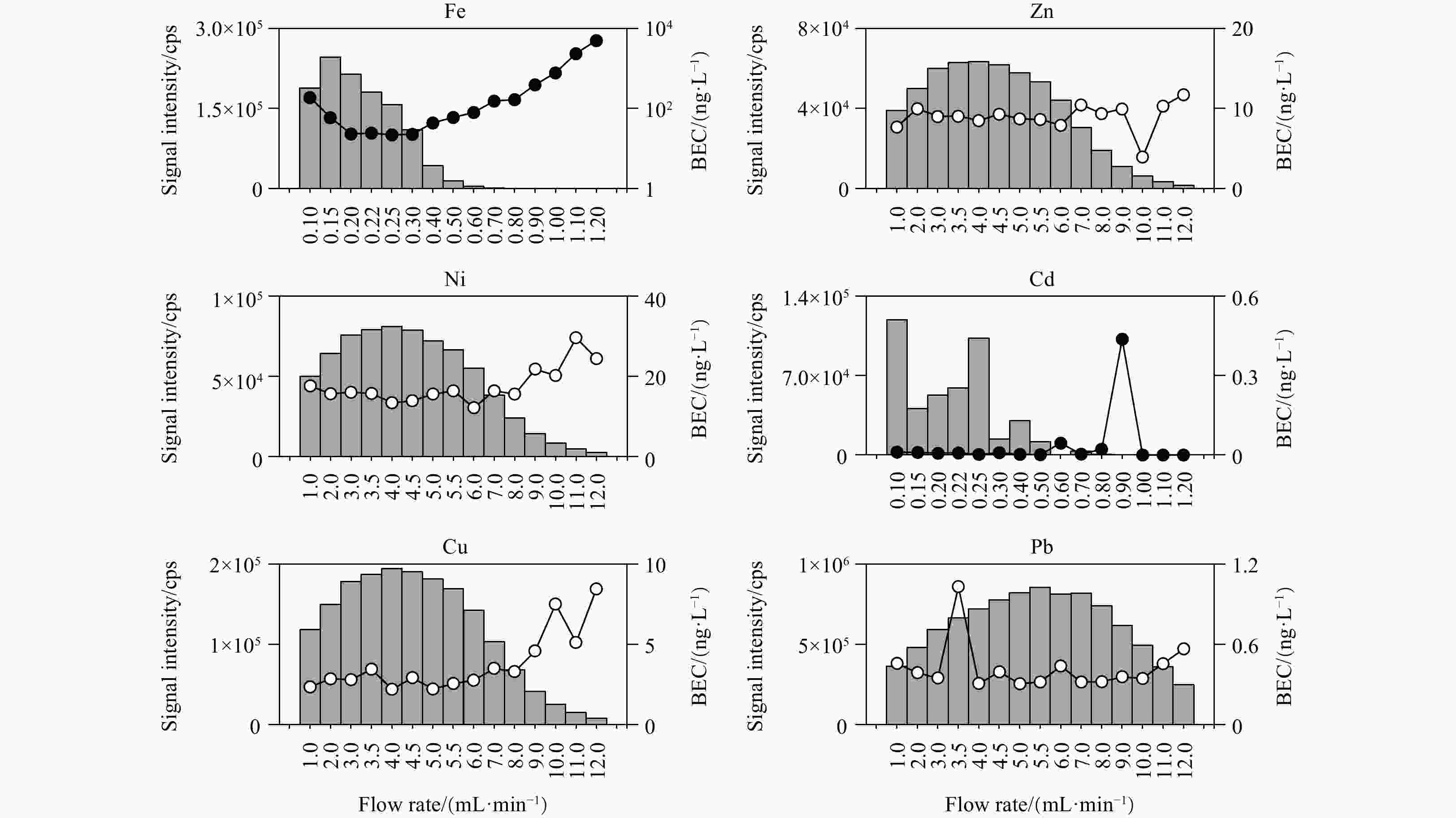
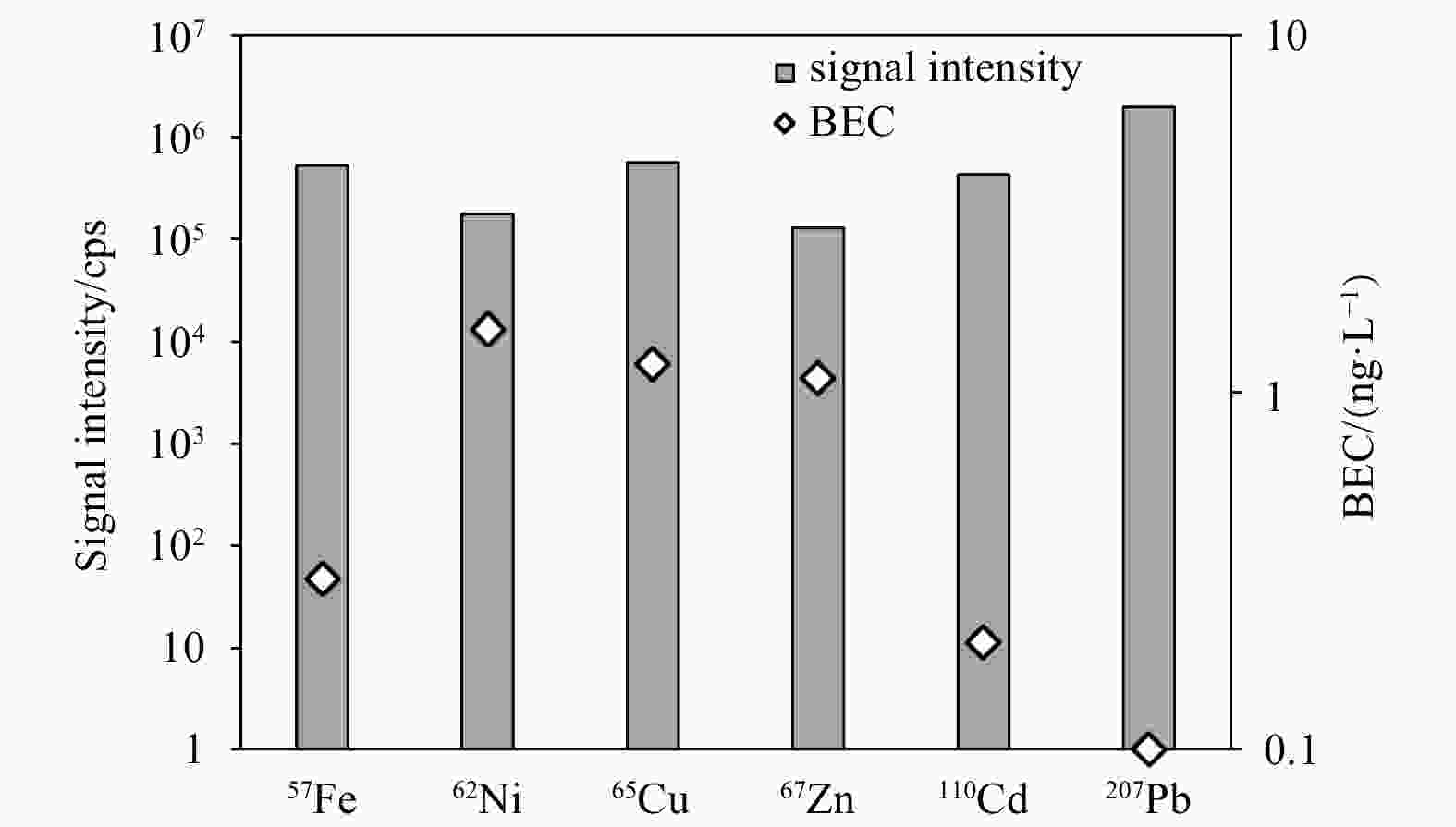
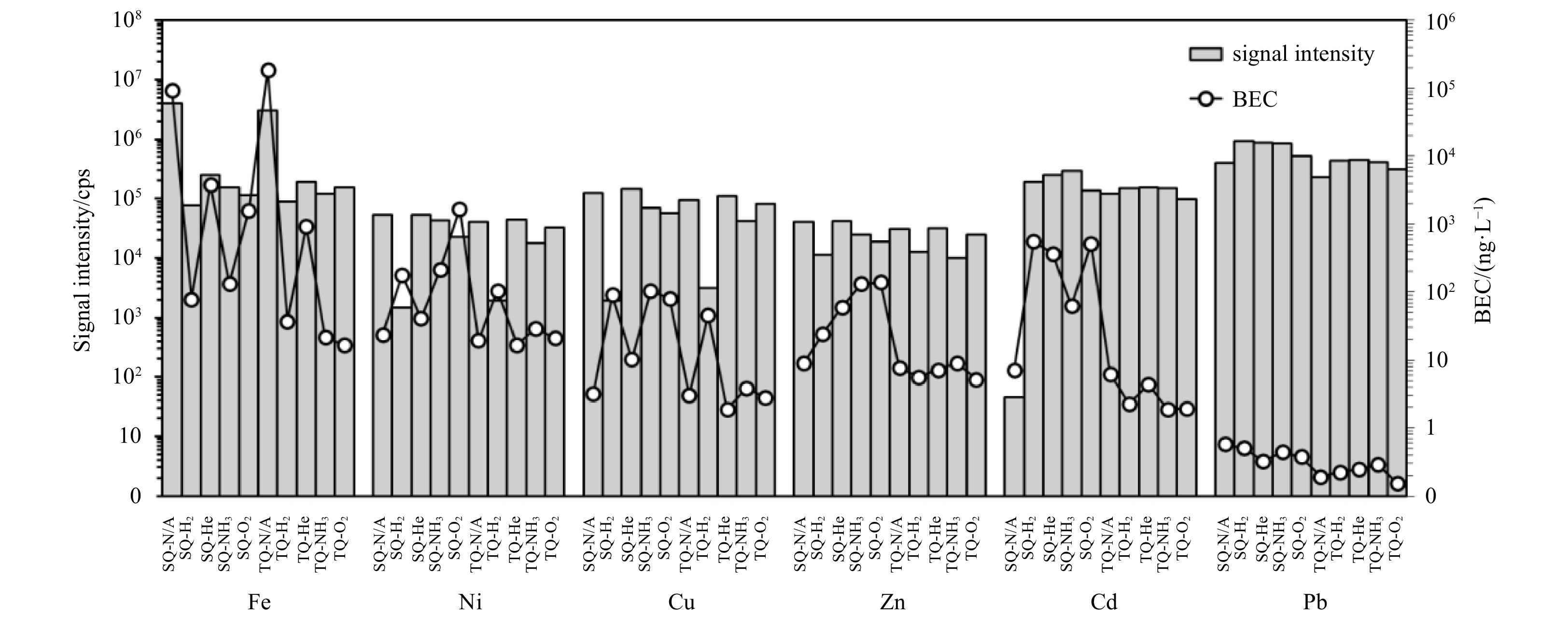
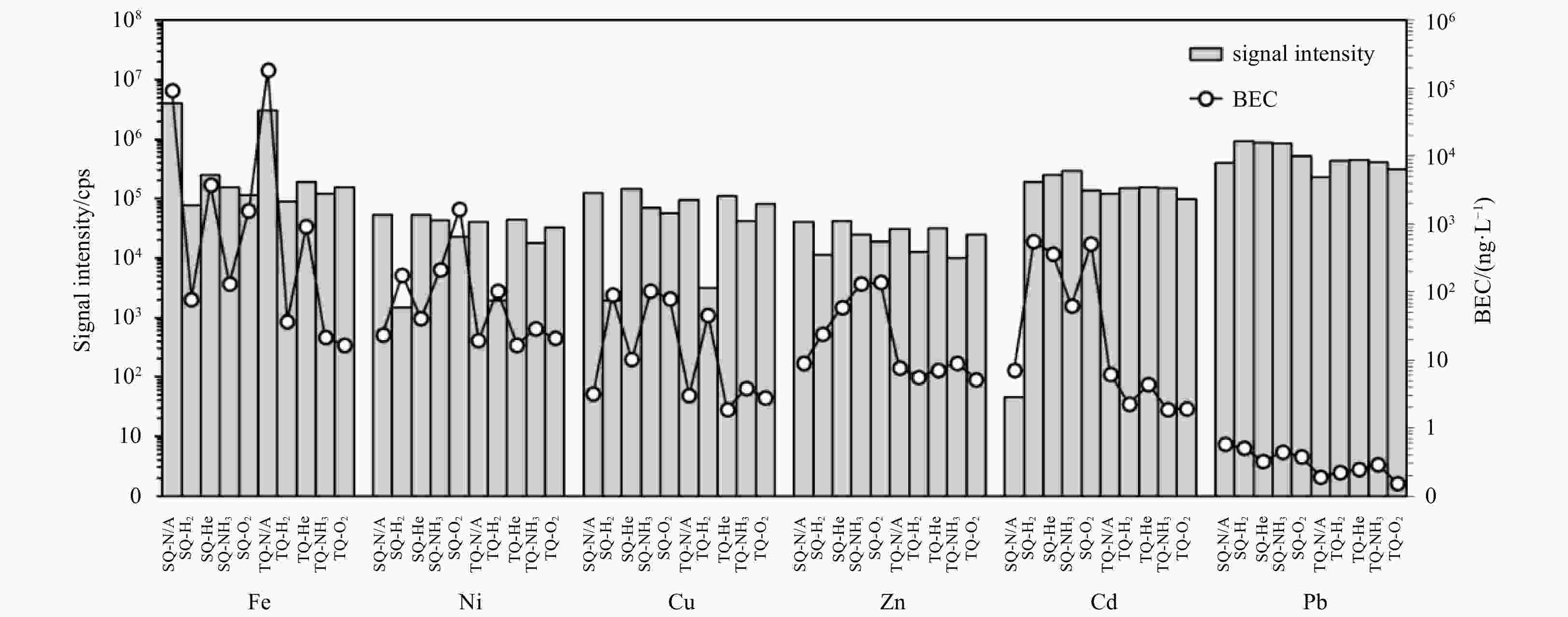
Figure 1. Signal intensity and background equivalent concentration (BEC) of each element analyzed using ten different combinations of collision/reaction cell gases and single-quad/triple-quad modes. All modes are auto-tuned by the instrument before the experiment. N/A represents no gas; the cps means counts per second.
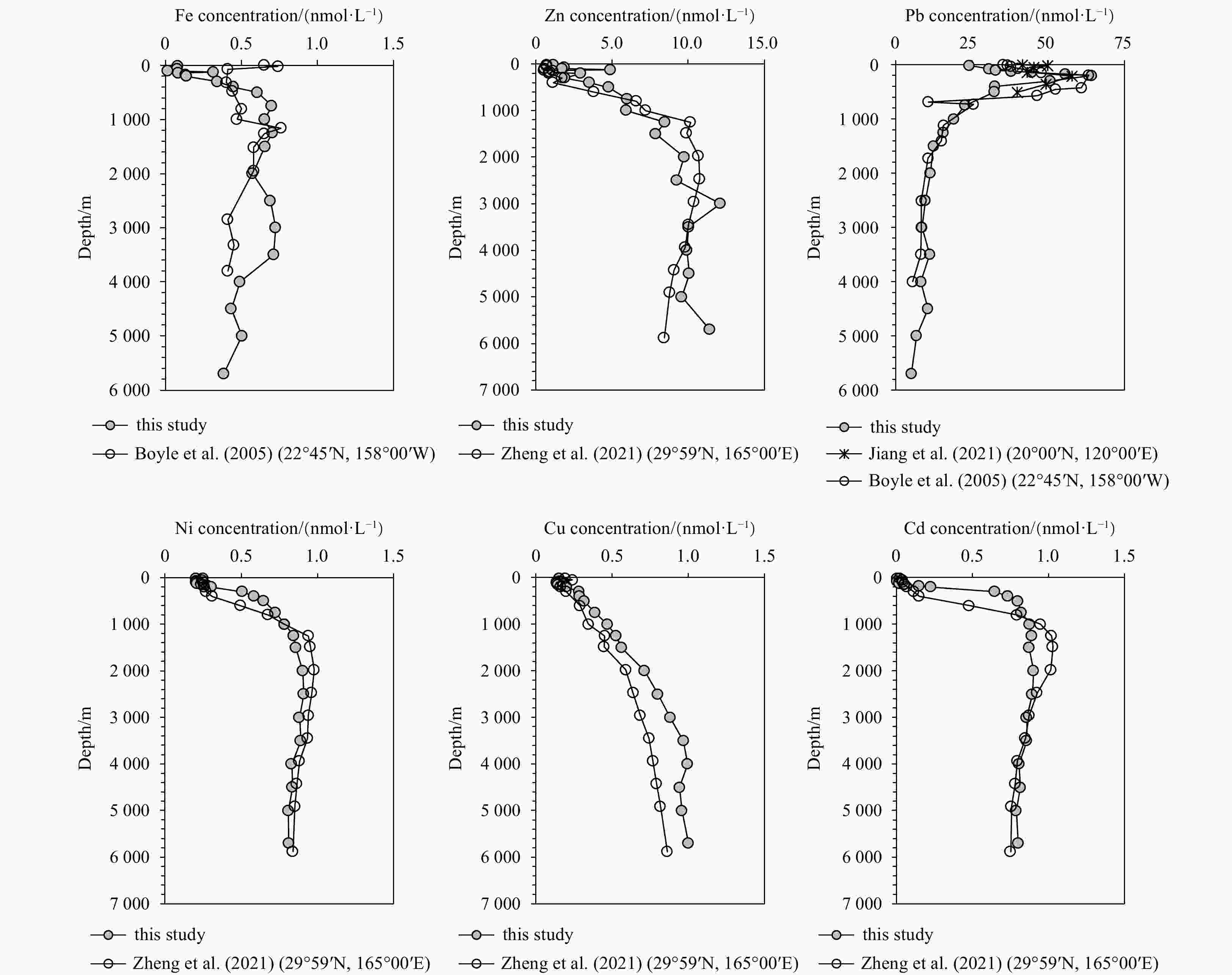
Figure 4. Vertical profiles of dissolved Fe, Zn, Pb, Ni, Cu and Cd in the subtropical Northwest Pacific (GEOTRACES GP09; Station K9, 11°00′N, 149°50′E). The comparison profiles were taken from the nearby GEOTRACES cross station (29°59′N, 165°00′E) for Zn, Ni, Cu, and Cd (Zheng et al., 2021), Fe and Pb were from the previous literature in the North Pacific reported by Boyle et al. (2005) and Jiang et al. (2021).
Table 1. ICP-MS/MS operating conditions
Parameter Operating condition Focus lens/V He mode: 1.25
NH3 mode: –0.5D1 lens/V –350 D2 lens/V He mode: –148
NH3 mode: –147.5CRC gas flow/(mL·min−1) He mode: 4.0
NH3 mode: 0.22Deflection lens/V –30 Spray chamber temperature/°C 2.7 Peristaltic pump speed/(r·min−1) 40 Cool flow/(L·min−1) 14 Sampling depth/mm 5 Plasma power/W 1550 Auxiliary flow/(L·min−1) 0.8 Extraction lens/V –120 Nebulizer flow/(L·min−1) 1.08 Note: CRC is abbreviation of collision/reaction cell. Table 2. Procedure concentration blanks (nmol/L) and detection limits of this method
Elements Procedure blank Method detection limits Fe 0.13 0.028 Ni 0.03 0.002 3 Cu 0.006 8 0.003 3 Zn 0.037 0.024 Cd 0.29 0.13 Pb 0.42 0.29 Table 3. Fe, Ni, Cu, Zn, Cd and Pb concentrations analysis of standard materials
GSC, n=10 NASS-7, n=10 CASS-6, n=10 Measured value/(nmol·L−1) Consensus value/(nmol·L−1) Measured value/(μg·L−1) Consensus value/(μg·L−1) Measured value/(μg·L−1) Consensus value/(μg·L−1) Fe 1.554±0.115 1.535±0.115 0.337±0.029 0.351±0.026 1.60±0.09 1.56±0.12 Ni 4.261±0.024 4.393±0.205 0.244±0.007 0.248±0.018 0.420±0.002 0.418±0.040 Cu 1.240±0.024 1.099±0.149 0.198±0.004 0.199±0.001 0.541±0.003 0.530±0.032 Zn 1.498±0.024 1.433±0.103 0.44±0.01 0.42±0.08 1.30±0.09 1.27±0.18 Cd 0.386±0.004 0.364±0.022 0.016 3±0.000 3 0.016 1±0.001 6 0.021 3±0.000 8 0.021 7±0.001 8 Pb 0.041±0.001 0.039±0.004 0.002 9±0.000 1 0.002 6±0.000 8 0.010 7±0.000 9 0.010 6±0.004 0 SLEW-3, n=10 SLRs-6, n=10 Measured value/(μg·L−1) Consensus value/(μg·L−1) Measured value/(μg·L−1) Consensus value/(μg·L−1) Fe 0.595±0.012 0.569±0.059 86.3±1.1 84.3±3.6 Ni 1.31±0.01 1.23±0.07 0.619±0.044 0.616±0.022 Cu 1.54±0.07 1.55±0.12 22.4±0.3 23.9±1.8 Zn 0.165±0.006 0.201±0.037 1.80±0.07 1.76±0.12 Cd 0.049±0.001 0.048±0.004 0.006 3±0.002 2 0.006 3±0.001 4 Pb 0.008±0.000 0.009 0±0.001 4 0.180±0.002 0.17±0.026 Note: GSC is the abbreviation of GEOTRACES intercalibration samples. -
[1] Almeida C, Vasconcelos M, Barbaste M, et al. 2002. ICP-MS multi-element analysis of wine samples—a comparative study of the methodologies used in two laboratories. Analytical and Bioanalytical Chemistry, 374(2): 314–322. doi: 10.1007/s00216-002-1467-8 [2] AlSuhaimi A O, AlRadaddi S M, Ali A K A S, et al. 2019. Silica-based chelating resin bearing dual 8-Hydroxyquinoline moieties and its applications for solid phase extraction of trace metals from seawater prior to their analysis by ICP-MS. Arabian Journal of Chemistry, 12(3): 360–369. doi: 10.1016/j.arabjc.2017.10.006 [3] An B, Can S Z, Bakırdere S. 2020. Traceable and accurate quantification of iron in seawater using isotope dilution calibration strategies by triple quadrupole ICP-MS/MS: characterization measurements of iron in a candidate seawater CRM. Talanta, 209: 120503. doi: 10.1016/j.talanta.2019.120503 [4] Behrens M K, Muratli J, Pradoux C, et al. 2016. Rapid and precise analysis of rare earth elements in small volumes of seawater—Method and intercomparison. Marine Chemistry, 186: 110–120. doi: 10.1016/j.marchem.2016.08.006 [5] Bianchi F, Careri M, Maffini M, et al. 2003. Use of experimental design for optimisation of the cold plasma ICP-MS determination of lithium, aluminum and iron in soft drinks and alcoholic beverages. Rapid Communications in Mass Spectrometry, 17(3): 251–256. doi: 10.1002/rcm.907 [6] Biller D V, Bruland K W. 2012. Analysis of Mn, Fe, Co, Ni, Cu, Zn, Cd, and Pb in seawater using the Nobias-chelate PA1 resin and magnetic sector inductively coupled plasma mass spectrometry (ICP-MS). Marine Chemistry, 130–131: 12–20 [7] Boyle E A, Bergquist B A, Kayser R A, et al. 2005. Iron, manganese, and lead at Hawaii Ocean Time-series Station ALOHA: temporal variability and an intermediate water hydrothermal plume. Geochimica et Cosmochimica Acta, 69(4): 933–952. doi: 10.1016/j.gca.2004.07.034 [8] de Jong J, Schoemann V, Lannuzel D, et al. 2008. High-accuracy determination of iron in seawater by isotope dilution multiple collector inductively coupled plasma mass spectrometry (ID-MC-ICP-MS) using nitrilotriacetic acid chelating resin for pre-concentration and matrix separation. Analytica Chimica Acta, 623(2): 126–139. doi: 10.1016/j.aca.2008.06.013 [9] Evans E H, Clough R. 2005. Isotope dilution analysis. In: Worsfold P, Townshend A, eds. Encyclopedia of Analytical Science. 2nd ed. Amsterdam: Elsevier, 545–553 [10] Firdaus M L, Norisuye K, Sato T, et al. 2007. Preconcentration of Zr, Hf, Nb, Ta and W in seawater using solid-phase extraction on TSK-8-hydroxyquinoline resin and determination by inductively coupled plasma-mass spectrometry. Analytica Chimica Acta, 583(2): 296–302. doi: 10.1016/j.aca.2006.10.033 [11] Freslon N, Bayon G, Birot D, et al. 2011. Determination of rare earth elements and other trace elements (Y, Mn, Co, Cr) in seawater using Tm addition and Mg(OH)2 co-precipitation. Talanta, 85(1): 582–587. doi: 10.1016/j.talanta.2011.04.023 [12] He Man, Huang Lijin, Zhao Bingshan, et al. 2017. Advanced functional materials in solid phase extraction for ICP-MS determination of trace elements and their species—A review. Analytica Chimica Acta, 973: 1–24. doi: 10.1016/j.aca.2017.03.047 [13] Hwang T J, Jiang S J. 1997. Determination of trace amounts of zinc in water samples by flow injection isotope dilution inductively coupled plasma mass spectrometry. Analyst, 122(3): 233–237. doi: 10.1039/a606577e [14] Jackson S L, Spence J, Janssen D J, et al. 2018. Determination of Mn, Fe, Ni, Cu, Zn, Cd and Pb in seawater using offline extraction and triple quadrupole ICP-MS/MS. Journal of Analytical Atomic Spectrometry, 33(2): 304–313. doi: 10.1039/C7JA00237H [15] Janssen D J, Sieber M, Ellwood M J, et al. 2020. Trace metal and nutrient dynamics across broad biogeochemical gradients in the Indian and Pacific sectors of the Southern Ocean. Marine Chemistry, 221: 103773. doi: 10.1016/j.marchem.2020.103773 [16] Jensen L T, Wyatt N J, Twining B S, et al. 2019. Biogeochemical cycling of dissolved zinc in the western Arctic (Arctic GEOTRACES GN01). Global Biogeochemical Cycles, 33(3): 343–369. doi: 10.1029/2018GB005975 [17] Jiang Shuo, Zhang Jing, Zhang Ruifeng, et al. 2018. Dissolved lead in the East China Sea with implications for impacts of marginal seas on the open ocean through cross-shelf exchange. Journal of Geophysical Research, 123(8): 6004–6018. doi: 10.1029/2018JC013955 [18] Jiang Shuo, Zhang Jing, Zhou Hui, et al. 2021. Concentration of dissolved lead in the upper northwestern Pacific Ocean. Chemical Geology, 577: 120275. doi: 10.1016/j.chemgeo.2021.120275 [19] Kato T, Nakamura S, Morita M. 1990. Determination of nickel, copper, zinc, silver, cadmium and lead in seawater by isotope dilution inductively coupled plasma mass spectrometry. Analytical Sciences, 6(4): 623–626. doi: 10.2116/analsci.6.623 [20] Komjarova I, Blust R. 2006. Comparison of liquid-liquid extraction, solid-phase extraction and co-precipitation preconcentration methods for the determination of cadmium, copper, nickel, lead and zinc in seawater. Analytica Chimica Acta, 576(2): 221–228. doi: 10.1016/j.aca.2006.06.002 [21] Kuma K, Isoda Y, Nakabayashi S. 2003. Control on dissolved iron concentrations in deep waters in the western North Pacific: Iron(III) hydroxide solubility. Journal of Geophysical Research, 108(C9): 3289. doi: 10.1029/2002JC001481 [22] Lee J M, Boyle E A, Echegoyen-Sanz Y, et al. 2011. Analysis of trace metals (Cu, Cd, Pb, and Fe) in seawater using single batch nitrilotriacetate resin extraction and isotope dilution inductively coupled plasma mass spectrometry. Analytica Chimica Acta, 686(1−2): 93–101. doi: 10.1016/j.aca.2010.11.052 [23] Li Li, Wang Xiaojing, Liu Jihua, et al. 2017. Dissolved trace metal (Cu, Cd, Co, Ni, and Ag) distribution and Cu speciation in the southern Yellow Sea and Bohai Sea, China. Journal of Geophysical Research, 122(2): 1190–1205. doi: 10.1002/2016JC012500 [24] Liao Wen-Hsuan, Takano S, Yang Shun-Chung, et al. 2020. Zn isotope composition in the water column of the northwestern Pacific Ocean: the importance of external sources. Global Biogeochemical Cycles, 34(1): e2019GB006379 [25] Mackey D J, O’Sullivan J E, Watson R J, et al. 2002. Trace metals in the western Pacific: temporal and spatial variability in the concentrations of Cd, Cu, Mn and Ni. Deep-Sea Research Part I: Oceanographic Research Papers, 49(12): 2241–2259. doi: 10.1016/S0967-0637(02)00124-3 [26] Masson P, Vives A, Orignac D, et al. 2000. Influence of aerosol desolvation from the ultrasonic nebulizer on the matrix effect in axial view inductively coupled plasma atomic emission spectrometry. Journal of Analytical Atomic Spectrometry, 15(5): 543–547. doi: 10.1039/a909651e [27] Nishioka J, Obata H. 2017. Dissolved iron distribution in the western and central subarctic Pacific: HNLC water formation and biogeochemical processes. Limnology and Oceanography, 62(5): 2004–2022. doi: 10.1002/lno.10548 [28] Norisuye K, Ezoe M, Nakatsuka S, et al. 2007. Distribution of bioactive trace metals (Fe, Co, Ni, Cu, Zn and Cd) in the Sulu Sea and its adjacent seas. Deep-Sea Research Part II: Topical Studies in Oceanography, 54(1−2): 14–37. doi: 10.1016/j.dsr2.2006.04.019 [29] Pickhardt C, Izmer A V, Zoriy M V, et al. 2006. On-line isotope dilution in laser ablation inductively coupled plasma mass spectrometry using a microflow nebulizer inserted in the laser ablation chamber. International Journal of Mass Spectrometry, 248(3): 136–141. doi: 10.1016/j.ijms.2005.11.001 [30] Rapp I, Schlosser C, Rusiecka D, et al. 2017. Automated preconcentration of Fe, Zn, Cu, Ni, Cd, Pb, Co, and Mn in seawater with analysis using high-resolution sector field inductively-coupled plasma mass spectrometry. Analytica Chimica Acta, 976: 1–13. doi: 10.1016/j.aca.2017.05.008 [31] Rimskaya-Korsakova M N, Berezhnaya E D, Dubinin A V. 2017. Analysis of molybdenum, tungsten, and vanadium in surface water of the Atlantic Ocean using solid phase extraction with 8-hydroxyquinoline and ICP MS determination. Oceanology, 57(4): 530–538. doi: 10.1134/S0001437017040154 [32] Rottmann L, Heumann K G. 1994. Development of an on-line isotope dilution technique with HPLC/ICP-MS for the accurate determination of elemental species. Fresenius’ Journal of Analytical Chemistry, 350(4): 221–227 [33] Sohrin Y, Urushihara S, Nakatsuka S, et al. 2008. Multielemental determination of GEOTRACES key trace metals in seawater by ICPMS after preconcentration using an ethylenediaminetriacetic acid chelating resin. Analytical Chemistry, 80(16): 6267–6273. doi: 10.1021/ac800500f [34] Song Panshu, Wang Jun, Chao Jingbo, et al. 2019. Accurate determination of trace molybdenum in drinking water by isotope dilution inductively coupled plasma mass spectrometry. Analytical Sciences, 35(7): 807–809. doi: 10.2116/analsci.19N010 [35] Sunda W G. 2012. Feedback interactions between trace metal nutrients and phytoplankton in the ocean. Frontiers in Microbiology, 3: 304 [36] Trueman C N, Rodgers K J, McLellan I S, et al. 2019. Geochemistry| Inorganic. In: Worsfold P, Poole C F, Townshend A, et al., eds. Encyclopedia of Analytical Science. 3rd ed. Cambridge: Elsevier, 271–282 [37] Vassileva E, Wysocka I, Orani A M, et al. 2019. “Off-line preconcentration and inductively coupled plasma sector field mass spectrometry simultaneous determination of Cd, Co, Cu, Mn, Ni, Pb and Zn mass fractions in seawater: procedure validation. Spectrochimica Acta Part B: Atomic Spectroscopy, 153: 19–27. doi: 10.1016/j.sab.2019.01.001 [38] Vegueria S F J, Godoy J M, de Campos R C, et al. 2013. Trace element determination in seawater by ICP-MS using online, offline and bath procedures of preconcentration and matrix elimination. Microchemical Journal, 106: 121–128. doi: 10.1016/j.microc.2012.05.032 [39] Warnken K W, Tang Degui, Gill G A, et al. 2000. Performance optimization of a commercially available iminodiacetate resin for the determination of Mn, Ni, Cu, Cd and Pb by on-line preconcentration inductively coupled plasma-mass spectrometry. Analytica Chimica Acta, 423(2): 265–276. doi: 10.1016/S0003-2670(00)01137-5 [40] Wu Jingfeng. 2007. Determination of picomolar iron in seawater by double Mg(OH)2 precipitation isotope dilution high-resolution ICPMS. Marine Chemistry, 103(3–4): 370–381. doi: 10.1016/j.marchem.2006.10.006 [41] Wu Jingfeng, Rember R, Jin Meibin, et al. 2010. Isotopic evidence for the source of lead in the North Pacific abyssal water. Geochimica et Cosmochimica Acta, 74(16): 4629–4638. doi: 10.1016/j.gca.2010.05.017 [42] Wuttig K, Townsend A T, van der Merwe P, et al. 2019. Critical evaluation of a seaFAST system for the analysis of trace metals in marine samples. Talanta, 197: 653–668. doi: 10.1016/j.talanta.2019.01.047 [43] Yamada N. 2015. Kinetic energy discrimination in collision/reaction cell ICP-MS: theoretical review of principles and limitations. Spectrochimica Acta Part B: Atomic Spectroscopy, 110: 31–44. doi: 10.1016/j.sab.2015.05.008 [44] Zhang Ruifeng, Jensen L T, Fitzsimmons J N, et al. 2019a. Dissolved cadmium and cadmium stable isotopes in the western Arctic Ocean. Geochimica et Cosmochimica Acta, 258: 258–273. doi: 10.1016/j.gca.2019.05.028 [45] Zhang Ruifeng, Zhu Xunchi, Yang Chenghao, et al. 2019b. Distribution of dissolved iron in the Pearl River (Zhujiang) Estuary and the northern continental slope of the South China Sea. Deep-Sea Research Part II: Topical Studies in Oceanography, 167: 14–24. doi: 10.1016/j.dsr2.2018.12.006 [46] Zheng Linjie, Minami T, Konagaya W, et al. 2019. Distinct basin-scale-distributions of aluminum, manganese, cobalt, and lead in the North Pacific Ocean. Geochimica et Cosmochimica Acta, 254: 102–121. doi: 10.1016/j.gca.2019.03.038 [47] Zheng Linjie, Minami T, Takano S, et al. 2021. Sectional distribution patterns of Cd, Ni, Zn, and Cu in the North Pacific Ocean: relationships to nutrients and importance of scavenging. Global Biogeochemical Cycles, 35(7): e2020GB006558 -




 下载:
下载: