Evidence of return of chum salmon released from Tangwang River by strontium marking method
-
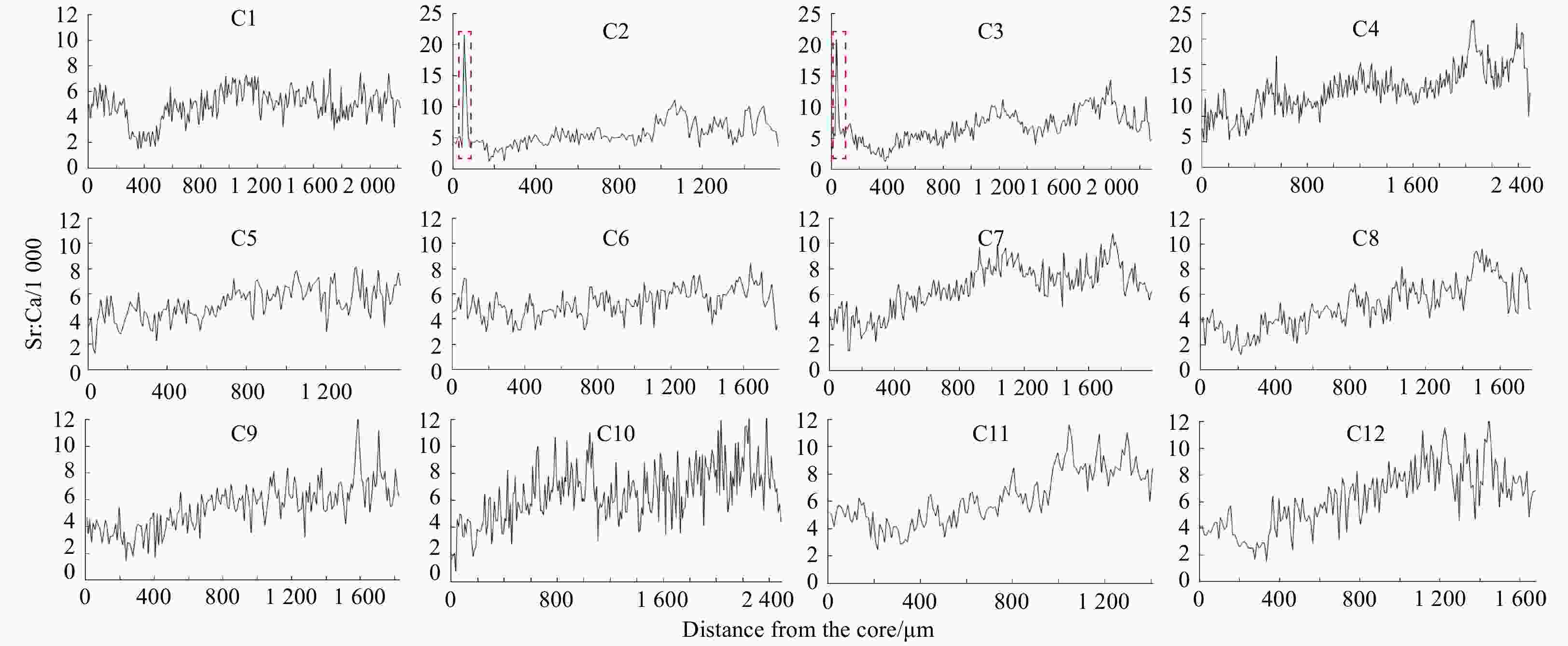
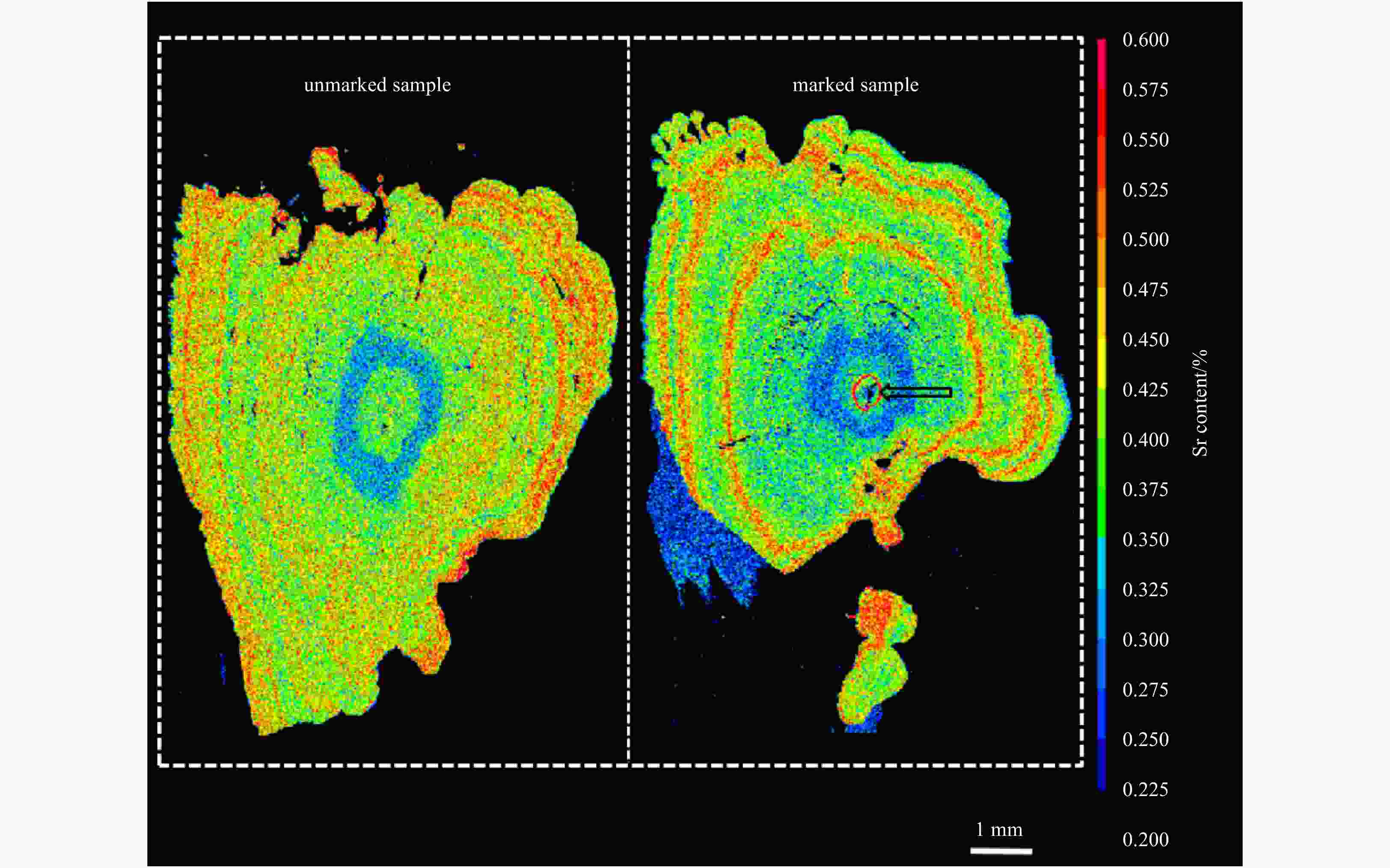
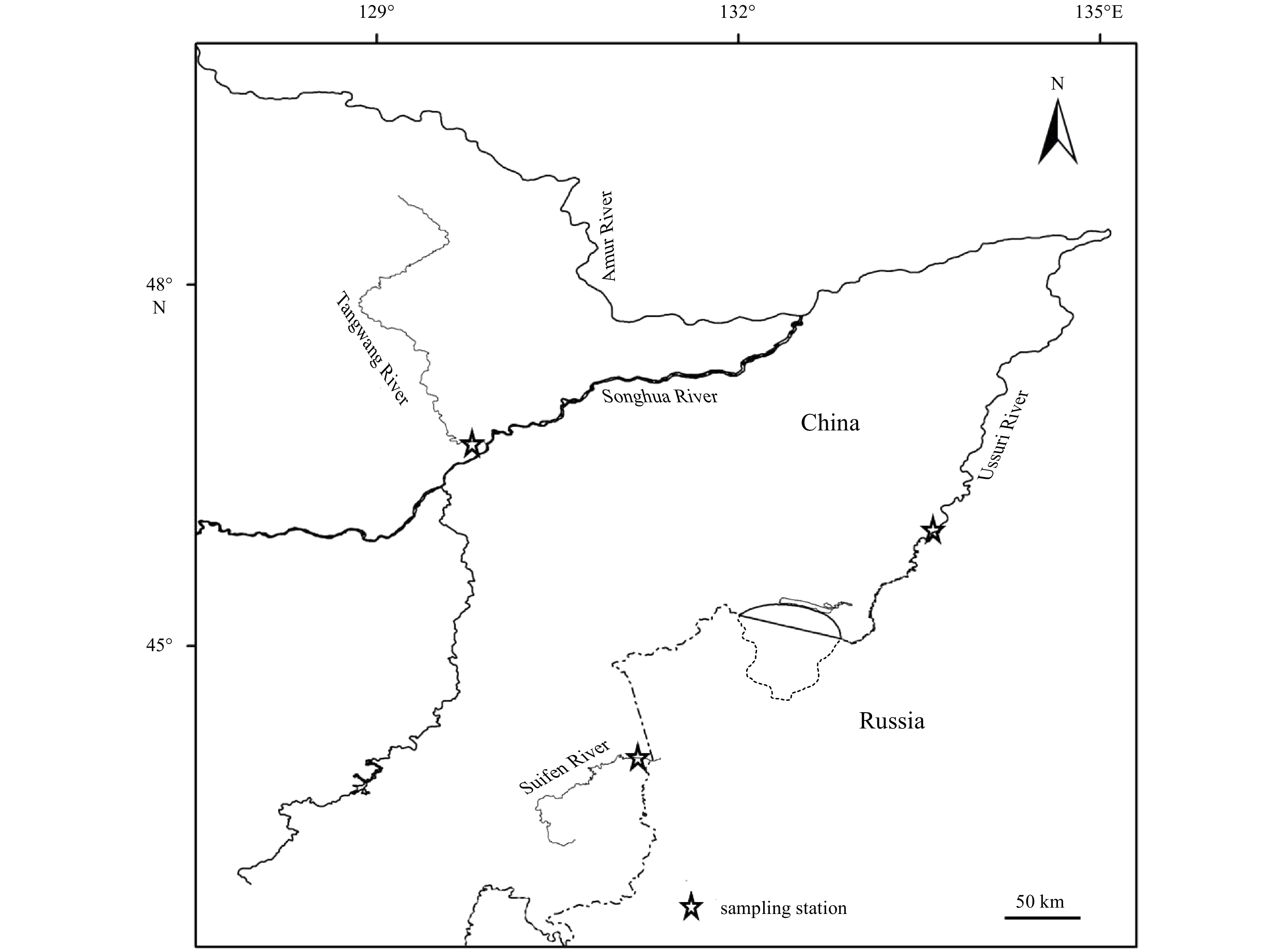
Abstract: In order to assess the effect of enhancement release of chum salmon (Oncorhynchus keta), otolith strontium (Sr) marking method was used to tag chum salmon released in Tangwang River in 2016. The homing chum salmon were detected and the samples were collected in Tangwang River, Ussuri River and Suifen River in the autumn of 2018. The samples were analyzed by examining Sr and calcium (Ca) fingerprints in the otolith using electron probe microanalysis. The results suggested that two samples collected in Tangwang River had the marking ring near the core of otolith where the Sr concentration and Sr/Ca ratio were significantly higher than comparative samples. Proving that the two fish belonged to the released population in Tangwang River in 2016. This article indicated the success of the enhancement release of chum salmon from the Tangwang River for the first time and also confirmed the validity of Sr marking in enhancement release of fishes.
-
Key words:
- chum salmon (Oncorhynchus keta) /
- otolith /
- strontium (Sr) marking /
- enhancement release
-
Table 1. The basic information of chum salmon samples
Samples Sex Body fork length/cm Body weight/g Age Sampling river C1 Female 65.5 2 859.5 3+ Tangwang River C2 Female 58.8 2 385.2 2+ Tangwang River C3 Male 48.9 1 019.7 2+ Tangwang River C4 Female 58.2 1 807.6 3+ Tangwang River C5 Male 55.1 1 703.7 2+ Ussuri River C6 Female 66.2 3 013.6 4+ Ussuri River C7 Female 69.8 3 902.5 4+ Ussuri River C8 Male 71.9 3 012.3 5+ Ussuri River C9 Male 46.3 1 205.9 1+ Suifen River C10 Male 44.9 1 098.2 1+ Suifen River C11 Male 49.2 1 322.5 2+ Suifen River C12 Female 54.9 1 761.6 2+ Suifen River -
[1] Arai T, Goto A, Miyazaki N. 2003a. Use of otolith microchemistry to estimate the migratory history of the threespine stickleback, Gasterosteus aculeatus. Journal of the Marine Biological Association of the United Kingdom, 83(1): 223–230. doi: 10.1017/S0025315403007008h [2] Arai T, Hayano H, Asami H, et al. 2003b. Coexistence of anadromous and lacustrine life histories of the shirauo, Salangichthys microdon. Fisheries Oceanography, 12(2): 134–139. doi: 10.1046/j.1365-2419.2003.00226.x [3] Arai T, Hirata T, Takagi Y. 2007. Application of laser ablation ICPMS to trace the environmental history of chum salmon Oncorhynchus keta. Marine Environmental Research, 63(1): 55–66. doi: 10.1016/j.marenvres.2006.06.003 [4] Beamish R J, Neville C M, Sweeting R M, et al. 2007. A proposed life history strategy for the salmon louse, Lepeophtheirus salmonis in the subarctic Pacific. Aquaculture, 264(1–4): 428–440. doi: 10.1016/j.aquaculture.2006.12.039 [5] Christian E, Heidi K, Eric C, et al. 2013. Species and life history affect the utility of otolith chemical composition for determining natal stream of origin for pacific salmon. Transactions of the American Fisheries Society, 142(5): 1370–1380. doi: 10.1080/00028487.2013.811102 [6] Dou Shuozeng, Yokouchi K, Yu Xin, et al. 2012. The migratory history of anadromous and non-anadromous tapertail anchovy Coilia nasus in the Yangtze River Estuary revealed by the otolith Sr: Ca ratio. Environmental Biology of Fishes, 95(4): 481–490. doi: 10.1007/s10641-012-0042-1 [7] Farrell J, Campana S E. 1996. Regulation of calcium and strontium deposition on the otoliths of juvenile tilapia, Oreochromis niloticus. Comparative Biochemistry and Physiology Part A: Physiology, 115(2): 103–109. doi: 10.1016/0300-9629(96)00015-1 [8] Goto A, Arai T. 2003. Migratory histories of three types of Cottus pollux (small-egg, middle-egg, and large-egg types) as revealed by otolith microchemistry. Ichthyological Research, 50(1): 67–72. doi: 10.1007/s102280300009 [9] Kang S, Kim S, Telmer K, et al. 2014. Stock identification and life history interpretation using trace element signatures in salmon otoliths. Ocean Science Journal, 49(3): 201–210. doi: 10.1007/s12601-014-0020-y [10] Kotake A, Arai T, Ozawa T, et al. 2003. Variation in migratory history of Japanese eels, Anguilla japonica, collected in coastal waters of the Amakusa Islands, Japan, inferred from otolith Sr/Ca ratios. Marine Biology, 142(5): 849–854. doi: 10.1007/s00227-003-1016-9 [11] Kullmann B, Hempel M, Thiel R. 2018. Chemical marking of European glass eels Anguilla anguilla with alizarin red S and in combination with strontium: in situ evaluation of short-term salinity effects on survival and efficient mass-marking. Journal of Fish Biology, 92(1): 203–213. doi: 10.1111/jfb.13508 [12] Liu Xiaolan. 2011. Effect of the construction of Yichun Hongshan Hydroelectric Power Station on fish resources in Tangwang River. Environmental Science and Management (in Chinese), 36(7): 160–162 [13] Phillis C C, Ostrach D J, Ingram B L, et al. 2011. Evaluating otolith Sr/Ca as a tool for reconstructing estuarine habitat use. Canadian Journal of Fisheries and Aquatic Sciences, 68(2): 360–373. doi: 10.1139/F10-152 [14] Si Fei, Ren Jiangong, Wang Qinglin, et al. 2019. Strontium marking on otoliths of Paralichthys olivaceus based on immersion experiments. Journal of Fishery Sciences of China (in Chinese), 26(3): 534–545. doi: 10.3724/SP.J.1118.2019.18270 [15] Wang Chen, Liu Wei, Zhan Peirong, et al. 2015. Exogenous Sr2+ sedimentation on otolith of chum salmon embryos. Chinese Journal of Applied Ecology (in Chinese), 26(10): 3189–3194 [16] Yang Jian, Arai T, Liu Hongbo, et al. 2006. Reconstructing habitat use of Coilia mystus and Coilia ectenes of the Yangtze River estuary, and of Coilia ectenes of Taihu Lake, based on otolith strontium and calcium. Journal of Fish Biology, 69(4): 1120–1135. doi: 10.1111/j.1095-8649.2006.01186.x [17] Yang Fuyi, Yan Baixing, Wang Qiang, et al. 2015. Assessment of fish stocks in the lower reaches of the Songhua River. Wetland Science (in Chinese), 13(1): 87–97 [18] Zhang Yi, Jiang Yazhou, Xu Kaida, et al. 2018. Evaluation on effectiveness of marking juvenile Sparus macrocephalus otoliths with strontium. Marine Fisheries (in Chinese), 40(2): 171–178 -




 下载:
下载: